Holguin is consolidating its position as a leading center for the use of HeberFERON. A Cuban medication developed by the Center for Genetic Engineering and Biotechnology (CIGB) for the treatment of basal cell carcinoma of the skin. It has been used in the province since 2012 in clinical trials.
Katerinee Batista Ochoa, a specialist in Dermatology and coordinator of the HeberFERON Program in the province. Highlighted the achievements and the importance of having this medication available in Primary Health Care (PHC).
Since 2016, over a thousand patients have been treated with this product. With an 85 percent complete response rate. Meaning the tumor has completely disappeared. A 91 percent objective response rate has also been achieved, encompassing both those who achieved a complete response and those who achieved a partial response. This translates to improved quality of life for these patients. Avoiding complex surgeries and mutilations, and achieving excellent aesthetic results at the lesion site.
HeberFERON was included in the essential medicines list in 2016. Enabling full coverage in all 14 municipalities of Holguin. This means that patients no longer need to travel to large hospitals or institutes to receive treatment with this biotechnological product. Which contains a combination of recombinant alpha 2b and gamma interferons. Instead, they can access the medication in their own communities. Thus facilitating accessibility and improving the quality of life for those affected.
Batista Ochoa noted that several Holguin polyclinics are currently showing favorable results in its use. Including the Pedro Díaz Coello, José Ávila Serrano, René Ávila, and Mario Gutiérrez polyclinics. Which are recognized for providing this service to the population. “Thanks to the training of medical personnel, we have been able to offer an effective and accessible treatment.”
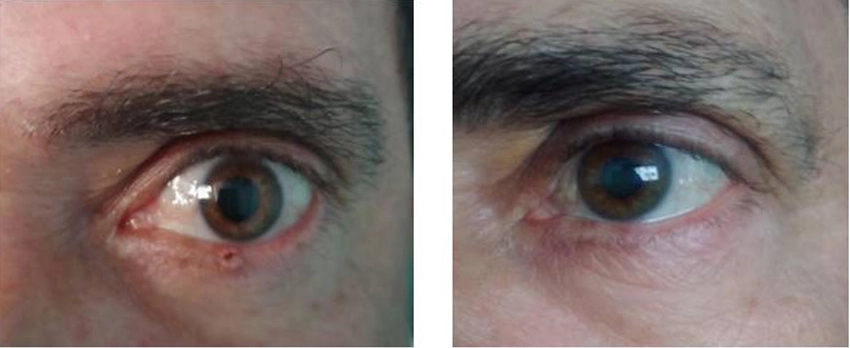
Example of the excellent aesthetic results it provides to patients after tumor removal. Photo: Courtesy of Dr. Iraldo Bello
Its use has shown positive effects in reducing several cancer cell lines. In many cases, after three weeks of treatment, the lesion disappears. With mild and transient adverse reactions. It has also been shown to have effects on other untreated lesions while reducing their recurrence rate. Resulting in very good aesthetic results after tumor removal.
In this regard, Dr. Iraldo Bello, a researcher at the Center for Genetic Engineering and Biotechnology (CIGB) and leader of the HeberFERON project. He stated that when HeberFERON is applied to a patient with basal cell carcinoma. So it not only provides treatment but also offers “hope, improved quality of life. And the opportunity to transform their future. Let us make our efforts a commitment to those who need it most.”
The results obtained using HeberFERON indicate that, in addition to being effective in reducing cancerous lesions. It has fewer side effects compared to traditional treatments. Its use in routine medical practice makes it possible to develop a comprehensive patient care model.
The progress made to date in localization is encouraging, and the possibility of evaluating its effectiveness in other types of cancer and dermatological conditions is being explored. HeberFERON is an example of how biotechnology can transform healthcare and improve patients’ quality of life.
- Women’s Health: A Call to Action - 28 de May de 2026
- Challenges of Living with Asthma from Childhood - 5 de May de 2026
- Resilience, the response of the health sector in Holguin this May 1st - 1 de May de 2026